
The use of animal free collagen could significantly lower the development costs of a medical device
The use of animal free recombinant collagen, rather than collagen extracted from animals, could significantly lower the development costs of a medical device by not contributing to the device regulatory classification so that the device is deemed Class II rather than an automatic Class III. Read the news release here: Mobile users click the link […]
Read More

Application Note: Electrospinning of Collagen
Electrospinning is an extremely versatile technique for the production of micro and nanofibres, as a consequence electrospun fibres have been fabricated for a wide range of applications within regenerative medicine including tissue engineering scaffolds and wound dressings. All these applications are seeking to replace or repair tissue architecture and functionality. Thus, collagen which is a […]
Read More

ProColl Achieves ISO 13485:2016 Medical Devices Quality Management System Compliance with UKAS Accreditation
ProColl has received certification from URS for their ISO 13485:2016 quality management system, with accreditation from the United Kingdom Accreditation Service (UKAS) for the scope of manufacture of ProColl’s bovine and recombinant collagen products. ISO 13485:2016 is the medical industry’s primary medical device quality management system standard that ensures compliance with regulatory and customer needs. […]
Read More

Innovate UK Transformative Technologies Grant – Engineering Biology
ProColl have been awarded an Innovate UK Transformative Technologies grant in engineering biology, for its precision fermentation. ProColl’s type II and III recombinant human collagen will be available to innovative technology users by the turn of the year. Contact us today for more information or to get a quote for our innovative materials. Read the […]
Read More

In vitro Comparison of ProColl’s Recombinant Human Collagen with Bovine Collagen
Human tissues are composed of multiple proteins and molecules that interact with one another to optimise tissue function. Collagen is a major component and interacts with the tissue environment as more than just a structural protein. Medical, pharmaceutical and aesthetics industries use collagen, where its structure provides mechanical robustness, and its interactions provide beneficial effects. […]
Read More

ProColl makes using structured collagen affordable at last!
We are now able to offer Bovine Acid Soluble Collagen, the go-to product for structured & biocompatible volume use, at a significantly lower cost to you than ever seen before. Our single figure price per gram compares with the market’s typically more than £2,000 per gram for native collagen. ProColl’s scale manufacturing ensures consistent collagen […]
Read More
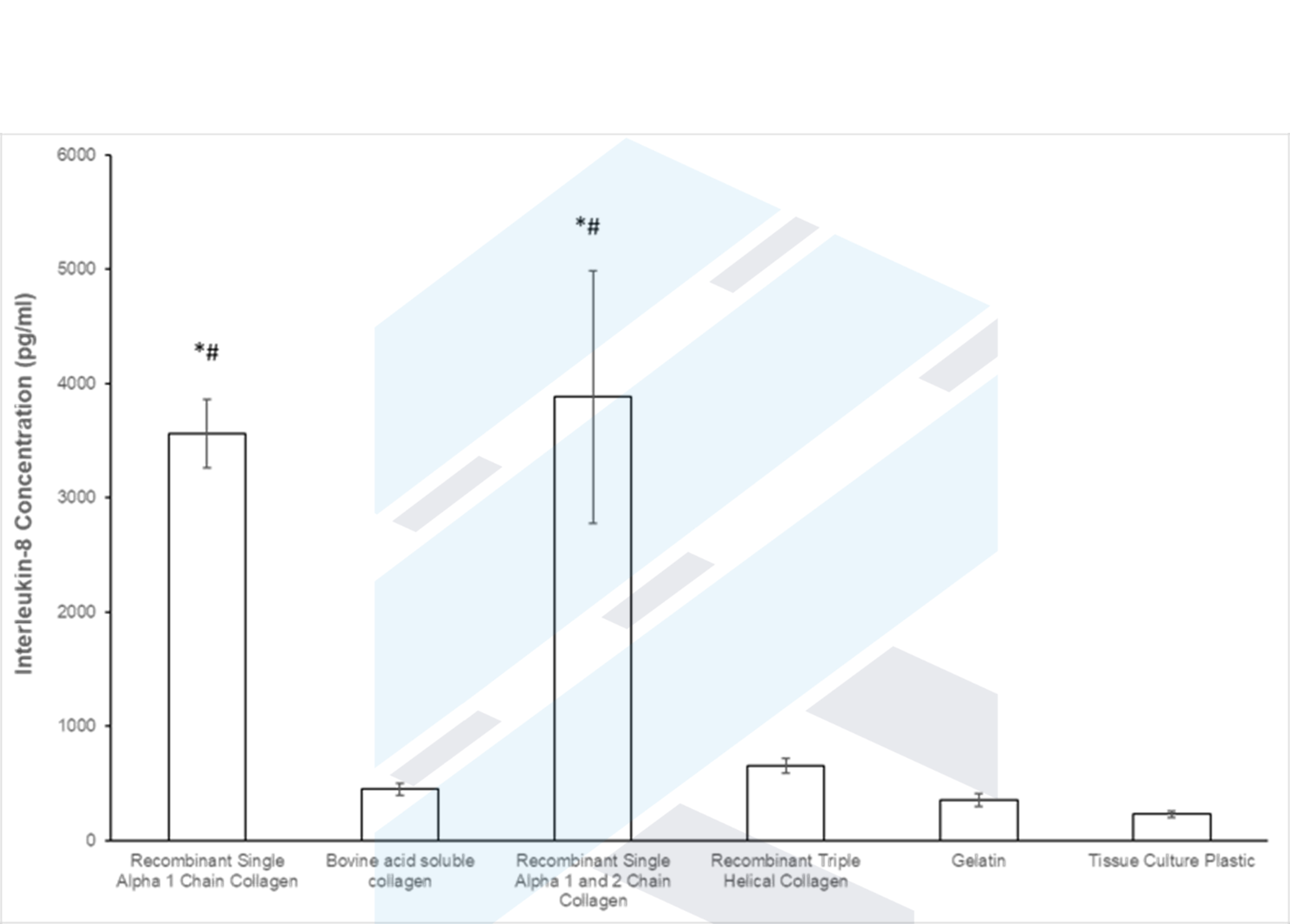
Collagen and Wound Healing – The Impact of Single Alpha Chain Collagen.
Collagen is key to restoring structure in wounds, given both its abundance as the most common protein in animals and innate interaction with various parts of the wound healing cascade. When collagen is damaged, individual chains that make up the triple helix become exposed which the body then identifies as needing repair. ProColl has a […]
Read More

Electrospinning Collagen Nanofibres: Retaining Native Structure and Avoiding Aggressive Solvents.
Electrospinning is a popular technique in tissue engineering and regenerative medicine to create biomimetic scaffolds, and the use of collagen for it’s bio-functionality is well described. However, it has become clear that the commonly used methods for the electrospinning of collagen produce fibres that lack the bio-functionality of native collagen, in part due to the […]
Read More
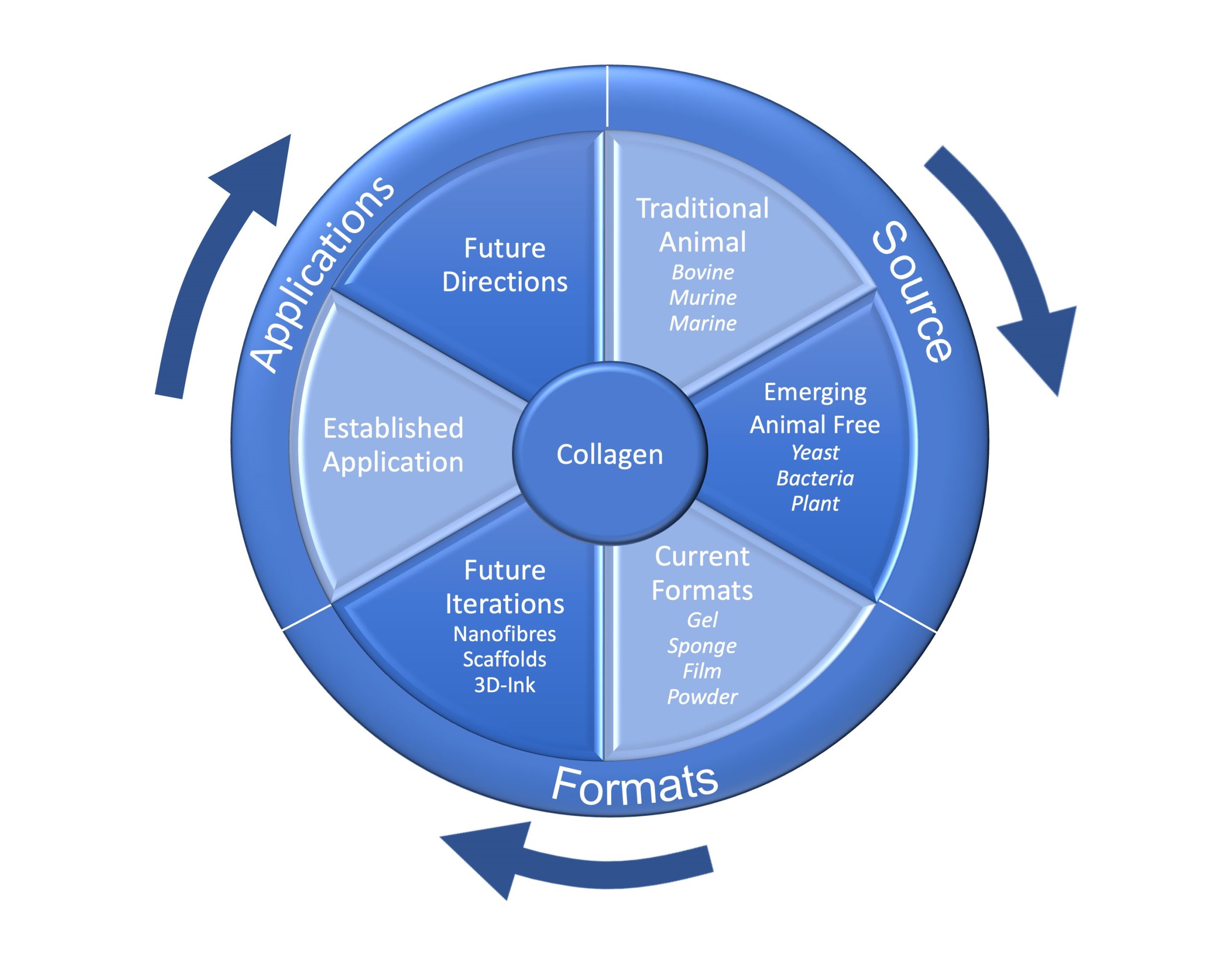
The World of Collagen – Poster
The World of Collagen. We present a pictorial overview of Collagen that demonstrates the far-reaching impact and potential of this important family of proteins. We see the supply of collagen, be that research-grade or for clinical applications moving from the established Type I bovine to recombinant synthetic biology forms and animal-free systems. ProColl is […]
Read More
Alternatives to scarce Matrigel and why they are needed.
Matrigel, first developed over 35 years ago has been used in 2D and 3D cell culture, however due to supply limitations, inconsistency of biochemical and mechanical properties and potential for antigenicity, researchers are now looking to other biomaterials for structural support of their cultures. This ProColl white paper examines the reasons for Matrigel’s limitations and […]
Read More
